Testimonials
Ti heat shields eliminated brake fade for my CTS-V
Essential for Production Race Cars

I wanted to say again that I'm very happy with the shims so far. At Pueblo the ambient temps were near ninety. Many cars in my run group were...
Read More ->
Essential for Production Race Cars

I remember the days in the 80s and 90s, when one of the key skills in driving a production race car was keeping from bending your brake pad backers. ...
Read More ->
Specials
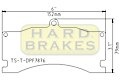
DPF7876 Titanium Brake Shim for Alcon, Mitsubishi EVO FQ
$97.99
Sale: $68.59
Special: 30% off
Sale: $68.59
Special: 30% off
Track Wisdom
What do aerospace engineer genius types think about these?
Wondering how our titanium alloy brake shims work to reduce your brake heat? Read on!
One of our (formerly skeptical) customers asked an aerospace engineer to give a simple explanation of how these shims work to reduce problems with brake heat. He has provided us with this copy of the engineer's response, which convinced him to try our heat shields. He is now a very satisfied Hard Brakes Customer
The Thermal Conductivity of a material often follows the electrical conductivity of that same material. So, any material that is good at conducting electricity will usually also be good at conducting and transferring heat. Aluminum and Copper are good examples of materials that are good at conducting electricity and heat. Dense metallic materials seem to be good at conducting heat and electricity. Less dense metallic material are usually not as good at conducting heat and electricity. And, less dense non-metallic materials (like phenolic) do not transfer heat or electricity very well.
So, if you have a short rod made out of phenolic or wood, you can hold one end of the rod and put the other end of the rod over a fire and the heat will not be transferred from the fire to your hand. Of course the end of the rod that is over the fire will eventually burn up. This is why the phenolic material on the break piston works the way it does. Phenolic will not transfer the heat from the pads to the pistons. But, the phenolic will eventually burn up. Or at least become brittle and disintegrate. Asbestos also has a very low coefficient of thermal conductivity and is not damaged by heat as much as phenolic.
If you hold the end of a short Aluminum rod over a fire, the heat will transfer up the rod very quickly and will burn your hand. The aluminum brake pistons do the same thing. The heat from the brakes and break pads is transferred through the aluminum piston to the break fluid.
Please read this article: http://phun.physics.virginia.edu/topics/thermal.html
Heat transfer engineers are always talking about thermal bridges and thermal breaks.
A thermal bridge is good at transferring heat, and a thermal break is not. Thermal breaks are made from elements or materials of low heat conductivity and are placed in an assembly to reduce or prevent the flow of heat between highly conductive materials. The insulation in the walls of your house is a thermal break. The wood handles on a cast iron skillet are thermal breaks.
This is going to sound silly, but please take a minute to understand that "Cold" is the absence of "Heat." We can not make "Cold." "Cold" happens naturally when we remove the "Heat."
I know this sounds silly, but it is true.
This may be easier to understand if we compare "heat and cold" to "light and dark."
"Dark" is the absence of "Light." We can not make "Dark." "Dark" happens naturally when we remove the "Light." So, if we want to brighten or otherwise make a room in our house bright, we turn on the "Light." When we leave the room and want to make the room "Dark" we do so by removing the "light." We turn off the light. We do not make a room dark by adding "Dark." We do not make a room dark by turning on the Dark. We make a room dark by removing Light.
Same thing applies to Heat. We do not make something Cold by adding "Cold." We make something Cold by removing "Heat." We can not make "Cold" but we sure can make "Heat."
Friction makes Heat. A lot of Friction makes a lot of Heat. The friction between the break pads and the break rotor makes a lot of Heat. And that heat must go somewhere, doesn't it. The heat is transferred to every component in the proximity of the area by conduction, convection and radiation. So, we need a "thermal brake" (Thermal barrier) between the break pads and the aluminum break pistons.
Compared to steel, Titanium is rugged, lightweight, strong, corrosion resistant and is only about 56 percent as dense as steel. Titanium also has a very high melting point of about 3100 degrees. This is much higher than the melting point of steel, which melts at a temp some 400 degrees less, and it is about 2000 degrees higher than aluminum. So, titanium is resistant to heat and seems to survive high temperatures better than most materials.
Titanium is NOT a good conductor of electricity. If the electrical conductivity of copper is considered to be 100%, then titanium would have an electrical conductivity of only 3.1%. Aluminum would be about 30%.
Titanium has a relatively low coefficient of thermal conductivity compared to other metallic materials.
The coefficient of thermal conductivity of Titanium is only about 10-22. Aluminum has a coefficient of thermal conductivity of about 220. So, Titanium should make a good thermal break or barrier.
Different materials coefficient of thermal conductivity
Air 0.025
Aluminum 220
Titanium 7.4-22.0 (Depending on alloy and temp)
[ Hard Brakes uses only 6AL/4V Titanium Alloy with a 7.4 conductivity ]
Steel 46
Stainless Steel 15
Copper 401
Concrete 1.0
Glass 1.1
Rubber 0.16
Wood 0.04-0.4
Cork 0.05
Conclusion:
Titanium seem to have all the properties that would make it a good, tough, heat resistant, resilient thermal insulator.
Stainless Steel 15
Copper 401
Concrete 1.0
Glass 1.1
Rubber 0.16
Wood 0.04-0.4
Cork 0.05
Conclusion:
Titanium seem to have all the properties that would make it a good, tough, heat resistant, resilient thermal insulator.